Lights, camera, action: engineering shape-shifting starfish cells

An optogenetic method for orchestrating the movement of starfish oocytes has been developed.
A research team from MIT (MA, USA), the University of Munich (Germany) and the Whitehead Institute for Biomedical Research (MA, USA) have developed an optogenetic method for controlling the shape of starfish egg cells in their earliest stages of development. This optical tool could help advance and guide the design of synthetic cells and life-like cellular functions for wound healing and drug delivery.
Starfish have long been used as classical models for understanding cell growth at various stages of development and as such, the key circuitry controlling motion and shape within starfish oocytes are well understood.
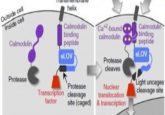
The enzyme GEF, which naturally circulates in starfish cell cytoplasm, is involved in this circuitry. Upon activation, GEF induces a change in the protein Rho – a key element in regulating cell mechanics – causing the protein to switch from a free-floating state to one that binds to the cell’s membrane. In this state, Rho triggers microscopic, muscle-like fibers to grow and spread across the membrane. These fibers twitch and enable the cell to contract, move and eventually divide.
Bioprinting and bioinks: the latest innovations in building synthetic biological structures
Discover new technologies and techniques that have broadened bioprinting’s applications from tissue engineering to drug discovery.
In previous work, corresponding author Nikta Fakhri’s group demonstrated that varying the concentrations of GEF could allow the cell’s movement to be manipulated. “This whole idea made us think whether it’s possible to hack this circuitry, to not just change a cell’s pattern of movements but get a desired mechanical response,” Fakhri explained.
With this in mind, the team applied established optogenetic techniques to genetically engineer the GEF enzyme to activate in response to light. mRNA from the genetically engineered enzyme could then be isolated and injected into starfish oocytes, enabling them to produce their own light-sensitive GEF. Each oocyte was then examined under a microscope and stimulated with different patterns and directions of light. Videos of the cell’s movement in response to the light were taken.
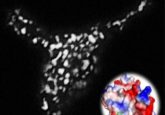
The team found that GEF was activated when light was aimed at specific sites of the cell, thereby recruiting Rho to the sites and triggering the growth of the muscle-like fibers. As a result, the fibers pulled or pinched the cell in these specific light-targeted sites. Through this method, the team was able to control the movement of the cells, and even direct various shape deformations.
“We realized this Rho-GEF circuitry is an excitable system, where a small, well-timed stimulus can trigger a large, all-or-nothing response,” Fakhri explained. “So, we can either illuminate the whole cell, or just a tiny place on the cell, such that enough enzyme is recruited to that region so the system gets kickstarted to contract or pinch on its own.”
Based on their observations, the team has developed a framework to predict and explain the mechanical responses to optogenetic stimuli. “This work provides a blueprint for designing ‘programmable’ synthetic cells, letting researchers orchestrate shape changes at will for future biomedical applications.” Concluded Fakhri.