Genetic elements that hijack bacterial viruses contribute to the spread of antibiotic resistance
A newly revealed mechanism of horizontal gene transfer improves understanding of how antibiotic resistance is transferred between bacteria.
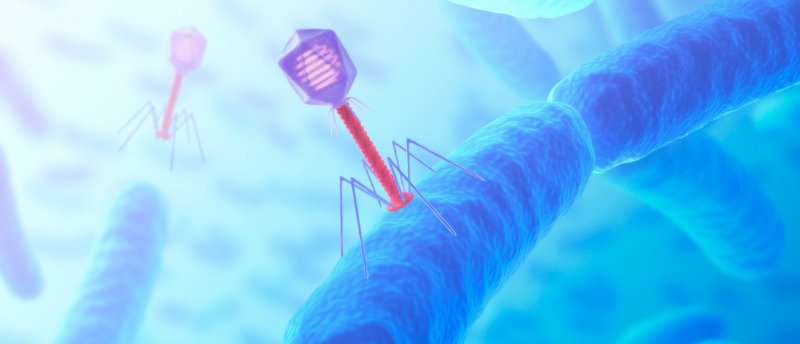
The increasing prevalence of antibiotic resistance (AR) is a critical threat to modern healthcare systems. AR genes are transferred between bacteria by horizontal gene transfer (HGT), a collective term that encompasses several distinct processes. One such process, transduction, relies on bacteriophage, viruses that infect bacteria, to transfer genetic material from one host cell to another. To better understand this process, a collaboration between researchers at the National University of Singapore (Queenstown, Singapore) and Imperial College London (UK) investigated Staphylococcus aureus pathogenicity islands (SaPIs), short, chromosomal islands that exploit the phage lifecycle for their mobilization.
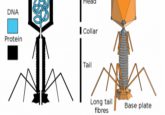
Phage infection occurs via two alternate cycles, initiated by the injection of phage DNA or RNA into the host. In the lytic cycle, this is translated to produce phage proteins that hijack the host replication, transcription and translation machineries for phage component production and genome replication. The phage genome contains pac sites, sequences recognized by phage small terminase, a protein that in turn recruits phage large terminase. This terminase complex packages DNA into phage heads during assembly of mature phages, which are released by host lysis. In the alternative lysogenic cycle, phage DNA instead integrates into the host chromosome to form a ‘prophage’. Prophages are dormant until host DNA-repair processes trigger their excision to begin the lytic cycle.
SaPI mobilization is normally prevented by a repressor protein, Stl, which binds an intergenic promotor to prevent excision from the chromosome. However, ‘helper’ prophages encode proteins that bind Stl and prevent repression of the SaPI promotor. Thus, helper prophage induction during the lysogenic cycle also initiates SaPI excision and replication. SaPIs encode their own small terminase, which recognizes the attachment site (attC), a sequence distinct from phage pac sequence. SaPI small terminase then recruits phage large terminase to facilitate packaging SaPI DNA into phage heads to form transducing particles.
Bacterial swimming machinery – a potential antibiotic target?
New mechanistic insights into the protein complex that powers the bacterial flagellum may assist antibiotic development.
It has previously been observed that SaPIs can mediate low-frequency generalized transduction, which is the mispackaging of host DNA containing sites with sequence similarity to the attC small terminase binding site.
The researchers of the current study created strains containing cadmium-resistance marker genes in the chromosomal DNA upstream and downstream of a SaPI. They observed that transfer of the markers downstream of the SaPI attC site occurred more frequently than upstream resistance markers by several orders of magnitude. The authors suggest that this provides evidence that SaPIs also cause lateral transduction (LT), a mechanism of transduction previously only described for prophage. LT arises when SaPI replication and terminate recruitment occurs prior to excision from the chromosome, packaging several phage headfuls of host DNA downstream of the SaPI attC site.
The authors also identified a novel process of SaPI transfer, lateral co-transduction (LcT), during which a SaPI switches from one strand to another during bidirectional replication, yielding two concatenated SaPI elements on a single strand. LcT from the attC site within the upstream SaPI therefore copies the entire downstream SaPI in addition to bacterial genes. Furthermore, unlike the requirement of phage LT for prophage induction, SaPI LT and LcT were evident during the lytic cycle. This previously undocumented mechanism has relevance to AR evolution because LT and LcT transfer much larger segments of bacterial DNA than other HGT mechanisms at high frequency, increasing the risk of virulence and AR gene transfer.
“In an unexpected twist of irony, our research has shown that parasites of the most prolific parasites on the planet (the phages) are probably the most powerful and efficient transducing agents currently known,” said co-corresponding author John Chen (National University of Singapore).
“This groundbreaking finding marks a paradigm shift in our understanding of bacterial evolution and will immensely influence the ways we combat antibiotic resistance,” added co-corresponding author José Penadésa (Imperial College London).