Take the plunge: cold-water acclimation affects cellular mechanisms
The cellular changes induced by cold water plunges have been revealed.
We’ve all heard about the potential health benefits of cold-water plunges, but what’s actually happening on a cellular level? Researchers in the Human and Environmental Physiology Research lab at the University of Ottawa (Canada) have investigated the effects of cold water acclimation on autophagic responses in young males. This work suggests cold-water acclimation can enhance cellular resilience against stress, with potential health and longevity implications.
Cold-water immersion is often used by athletes to reduce muscle soreness and inflammation post-exercise as well as improve circulation. This use of cold-water immersion is based on research that has investigated the changes in physiological mechanisms in response to cold acclimation, such as metabolic heat production, shivering and vasoconstriction. However, cold water immersion post resistance training has been shown to slightly attenuate muscle growth, highlighting that the increased recovery benefits do not come at no cost to the practitioner.
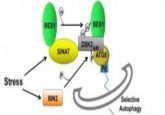
Previous research has shown that extreme cold can cause significant cellular damage, which is exacerbated with continued exposure to such cold temperatures. To prevent cell death in response to these conditions, cells initiate protective mechanisms, such as autophagy. Autophagy is the degradative process by which toxic protein aggregates and damaged organelles are broken down to prevent their accumulation and interruption of normal cellular function.
 Breaking the ice-repelling secrets of polar bear fur
Breaking the ice-repelling secrets of polar bear fur
Low ice adhesion properties of polar bear fur sebum offer sustainable solutions for preventing ice buildup on infrastructure.
Although physiological and cellular mechanistic changes in response to cold have been explored, few studies have evaluated the efficacy of cold acclimation on autophagy in animal models, with no known studies in humans. The current research team set out to fill this gap in our knowledge by examining the potential changes of in vivo autophagic responses and related pathways of cellular stress.
The study involved ten healthy males, between the ages of 20 and 26, being immersed in cold water at 57.2ºF (14oC) for 1 hour across seven consecutive days, with blood samples being collected before, at the midway point and after the 7-day acclimation period. In parallel to their in vivo study, the team exposed peripheral blood mononuclear cells isolated from blood samples taken prior to acclimation and 24hrs after the final cold water exposure, to additional ex vivo cooling to see if consistent cold exposure would attenuate damaging cellular responses in hypothermic conditions.
They found that autophagy was initially dysfunctional after high-intensity cold stress; however, consistent exposure over the 7-day period showed an increase in autophagic activity and decreased cellular damage signals. They also found that the cells’ cold tolerance post-acclimation was markedly improved.
“Our findings indicate that repeated cold exposure significantly improves autophagic function, a critical cellular protective mechanism,” commented senior author Glen Kenny. “This enhancement allows cells to better manage stress and could have important implications for health and longevity.”
The findings extend beyond ice baths for athletes and draw attention to the potential of cold-water acclimation in enhancing human health to prevent disease and slow aging on a cellular level. However, the team recognize that their study only applies to young males, and that they will need to conduct more studies with other cohorts to confirm that these cellular results are universal.